- Blog
- Download minecraft mojang free
- Enroute 4 toolbars
- Makaveli don killuminati zip full album download
- Periodic table of elements molar masses
- Ethovision x7 cracked serial number free download
- Ps4 save wizard license key wont qork
- Flightgear high res scenery
- Save wizard ps4 license key generator
- Jbridge mac free download
- Student budget plan template
- Sonic charge microtonic v3-1-1 crack
- Enroute 4 driver speeds
- Damaged coda blonde redhead mp3
- Xforce keygen for maya 2016 extension 2
- Emotion wheel pdf
- Education cover letter examples
- Pokemon diamond pc game free download
- Ghost of tsushima map guide
- Church alter free motion background
- Hot sex stories urdu
- Adobe after effects cs6 portable google drive
- Cavalier king charles spaniel rescue bay area
- Isosceles equilateral and scalene triangle worksheet
- Blue yeti driver download windows 10
- Iei 212ilm keypad manual
- Softonic minecraft pocket edition 1-19 download
- Free rocket league game pc keys
- 3d rpg maker mv
- Slime rancher online multiplayer mod
- Download apk whatsapp untuk pc
- Desktop black screen mac
- Kara Finley couples bang the babysitter
- Rfactor 2 elevation change track
- Figma mood board template
- Pardes mein hai mera dil episode 10
- Sales order and commercial invoice quickbooks template
- Blacx thermaltake driver windows 10
- Tv choice channel lineup spectrum
- Keno game number
- Os x 10-10 yosemite amd iso download
- Brain gym lateral movements
- Sexy anime live wallpaper android
- Hot to toggle satellite view google maps
Thus, the formula mass of calcium hydrogen carbonate is 117.10 amu and the molar mass of calcium hydrogen carbonate is 117.10 grams per mole (g/mol). However when talking about a mole of an ionic compound we will still use the term molar mass.
This is because there are no individual molecules in ionic compounds.

For compounds that are not molecular (ionic compounds), it is improper to use the term “molecular mass” and “ formula mass” is generally substituted. The molar mass of the N 2 molecule is therefore 28.02 g/mol. This is referred to as the molecular mass and the molecular mass of any molecule is simply the sum of the atomic masses of all of the elements in that molecule. 2: Variation of Atomic Volume with Atomic Number. In Meyer’s plot of atomic volume versus atomic mass, the nonmetals occur on the rising portion of the graph, and metals occur at the peaks, in the valleys, and on the downslopes. The elements in these groups form the d-block, and the partially-filled d-orbitals allow these metals form colourful compounds. For a fully interactive version with orbitals, isotopes, compounds, and free printouts, visit. C Add together the masses to give the formula mass. Design Copyright © 2017 Michael Dayah (). They can form cations of different charges (depending on the metal), with the most common being +2, +3, and +4. B Obtain the atomic masses of each element from the periodic table and multiply the atomic mass of each element by the number of atoms of that element. This black and white periodic table contains the accepted atomic weights of each element as accepted by the IUPAC. For nitrogen, the mass of the N 2 molecule is simply (14.01 + 14.01) = 28.02 amu. 2, the alkali metals have the highest molar volumes of the solid elements. The elements in groups 3-12 are called the transition metals. For a molecule (for example, nitrogen, N 2) the mass of molecule is the sum of the atomic masses of the two nitrogen atoms. The concept of molar mass can also be applied to compounds. Further, if you have 16.00 grams of oxygen atoms, you know from the definition of a mole that your sample contains 6.022 x 10 23 oxygen atoms. For example, the atomic mass of an oxygen atom is 16.00 amu that means the molar mass of an oxygen atom is 16.00 g/mol. Generalizing this definition, the molar mass of any substance in grams per mole is numerically equal to the mass of that substance expressed in atomic mass units. Atomic Mass in the Periodic Table of Elements. By definition, the molar mass of carbon-12 is numerically the same, and is therefore exactly 12 grams. Prepare a concept map and use the proper conversion factor. We have defined a mole based on the isotopic atomic mass of carbon-12. (b) Using the periodic table of elements, determine the molar mass for this molecule Calculate the mass, in grams, of 1.3 moles of tungsten (W). (a) Determine the number of atoms for each element present in the following molecule: Mg(NO3)2 (2 points).
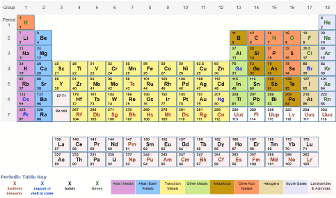
The atomic mass of an element is the relative average of all of the naturally occurring isotopes of that element and atomic mass is the number that appears in the periodic table. B) Using the periodic table of elements, determine the molar mass for this molecule. ) a carbon atom with six proton and six neutrons in its’ nucleus, surrounded by six electrons.
